The advantages of Supercritical CO2
Compared to the classic organic solvents used in extractive processes such as hexane, carbon dioxide also has other advantages such as:
- It is not toxic to either the workers or the surrounding environment.
- It has a low cost and can be easily released into the atmosphere, as it uses CO2 previously concentrated from the atmosphere, (so there are zero emissions).
- After solubilization of the compounds of interest, it can be easily reconverted to the gaseous state and eliminated or recirculated in the same process without further processing. This allows for a finished product with no trace of solvents.
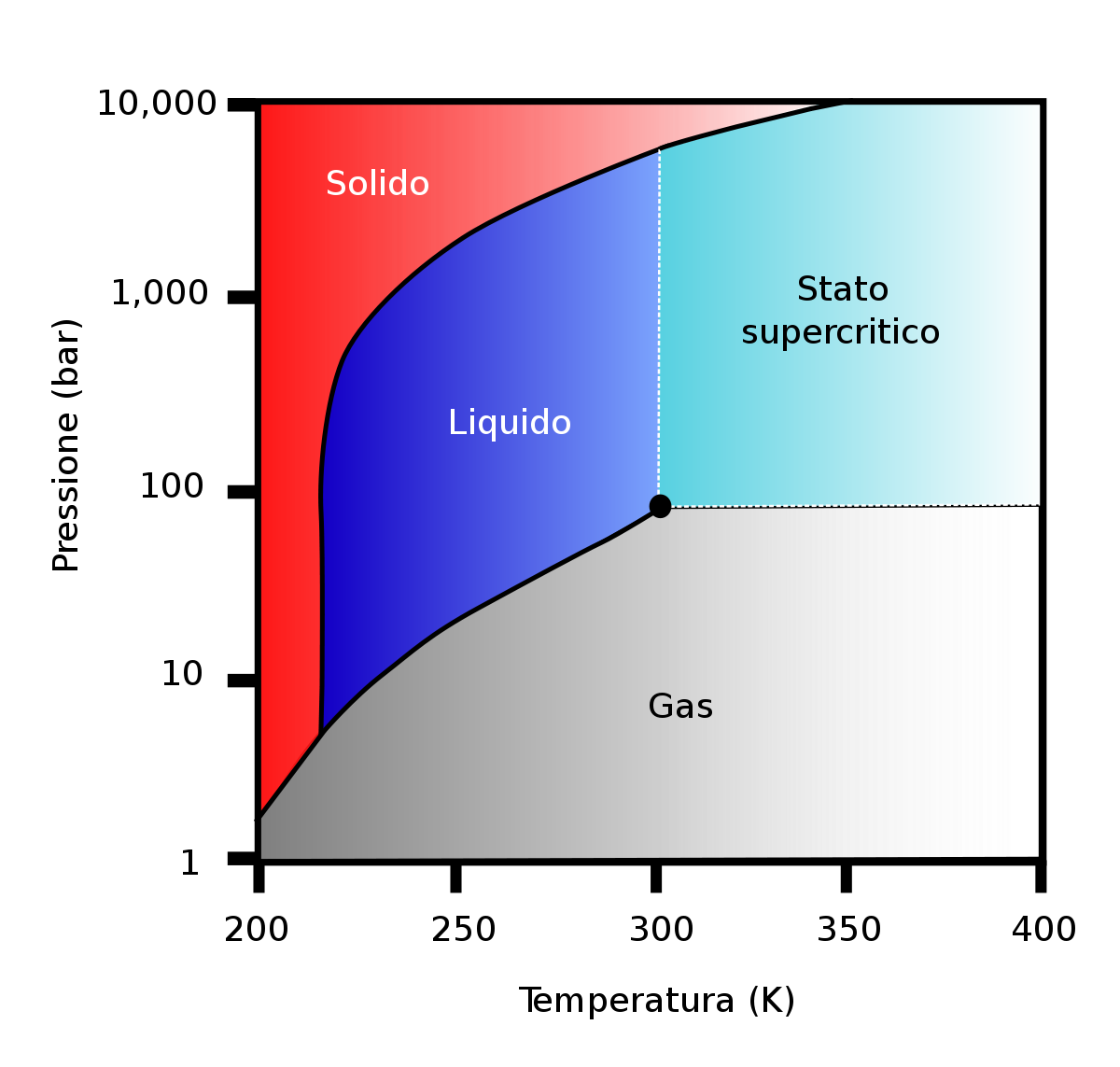
- A critical temperature (Tc) close to the environmental one reduces the damage of thermolabile substances and the final product is more valid and safe.
- Saturation with CO2 in the supercritical state of the extraction chamber briefly and in the absence of O2 will inhibit all chemical and biological oxidative processes that may alter the matrix or compounds to be extracted.
- Being a low molecular weight gas, any matrix will have a high permeability for it, and thus the extraction time will be shorter than that evaluated with other solvents.